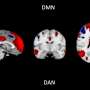
Researchers from Université Savoie Mont Blanc, Radboud University, and the University of Oxford have identified a potential biomarker for cognitive decline associated with Alzheimer’s disease. Their study reveals that changes in the brain’s intrinsic connectivity, specifically between the default mode network (DMN) and dorsal attention network (DAN), may serve as an independent fMRI marker for Alzheimer’s pathology. This discovery could provide valuable insights into the disease’s progression and improve diagnostic capabilities.
Current methods to diagnose Alzheimer’s lack a universally accepted biomarker that can pinpoint the onset of abnormal protein accumulation leading to cognitive decline. Understanding the large-scale functional breakdown in Alzheimer’s remains a complex challenge. Researchers have historically tied amyloid and tau pathology, vascular lesions, and neurodegeneration together, yet the relationship among these factors is not fully understood. Notably, beta-amyloid accumulation can begin years before clinical symptoms appear, supporting the notion that Alzheimer’s disease exists on a continuum from normal aging to dementia.
Dueling Networks in the Brain
Neurocognitive networks in the human brain can exhibit both cooperative and competitive behavior. Resting-state functional MRI (fMRI) has been instrumental in exploring how these networks engage without the influence of external tasks. The DMN is typically active during periods of self-reflection, while the DAN activates during focused tasks, such as sports or complex problem-solving.
Prior research has indicated that alterations in resting-state connectivity within and between these networks are linked to cognitive performance in healthy individuals. Alzheimer’s disease appears to disrupt these connectivity patterns, leading to cognitive decline. The concept of a “functional imbalance” in resting-state networks has emerged as a potential factor in neurodegenerative pathology.
The recent study, titled “The intrinsic connectivity between the default mode and dorsal attention networks is an independent fMRI biomarker of Alzheimer’s disease pathology burden,” published in NeuroImage, analyzed data from 182 participants in the Alzheimer’s Disease Neuroimaging Initiative (ADNI). Researchers examined MRI and PET scan data, focusing on the anticorrelation between DMN and DAN and its association with Alzheimer’s pathology, specifically amyloid and tau burdens.
Insights from Resting-State Connectivity
The analysis revealed that participants with high amyloid levels and cognitive impairment exhibited a reduced separation between the DMN and DAN signals. This diminished distinction suggests that the networks become less effective at maintaining focus, potentially complicating tasks that require concentration and memory retrieval. Overall, researchers found that weaker DMN-DAN anticorrelation correlated with lower cognitive performance scores.
Multivariate models that accounted for age, sex, education, and tau burden still indicated that DMN-DAN connectivity independently predicted cognitive performance. Approximately 5% of the variance in cognitive performance was linked to this specific fMRI measure. The study also found that the relationship extended to scores on the Mini-Mental State Examination and various cognitive tests from the ADNI neuropsychological battery, reinforcing the significance of DMN-DAN connectivity in understanding cognitive decline.
Interestingly, education level, often viewed as an indicator of cognitive reserve, did not alter this association, emphasizing the potential of DMN-DAN connectivity as a reliable marker of cognitive impairment.
The findings suggest a shift in understanding the mechanisms behind cognitive decline in Alzheimer’s disease. Rather than viewing changes in DMN-DAN connectivity as merely intermediaries in tau-dependent neurodegeneration, the authors propose a more complex interaction involving multiple factors, including vascular health, sleep quality, and stress.
Future research directions highlighted in the study include longitudinal and interventional studies aimed at determining whether network dysfunction occurs before or after abnormal protein accumulation. Researchers also propose developing personalized risk models that integrate normative DMN-DAN connectivity patterns, pathology measures, and lifestyle factors from extensive longitudinal datasets.
In summary, this study places a simple resting-state measure at the forefront of understanding Alzheimer’s disease. As research continues to evolve, these insights could lead to more effective diagnostic tools and interventions for those affected by this debilitating condition.