Vanda Pharmaceuticals (NASDAQ: VNDA) experienced a surge in its stock price during the premarket session on Wednesday following the U.S. Food and Drug Administration (FDA) approval of its new oral medication, Bysanti. This treatment is aimed at addressing bipolar I disorder and schizophrenia. Additionally, Vanda’s application for a drug targeting a rare skin condition is currently under FDA review, marking a pivotal moment for the company.
FDA Approval for Rare Skin Disease Drug
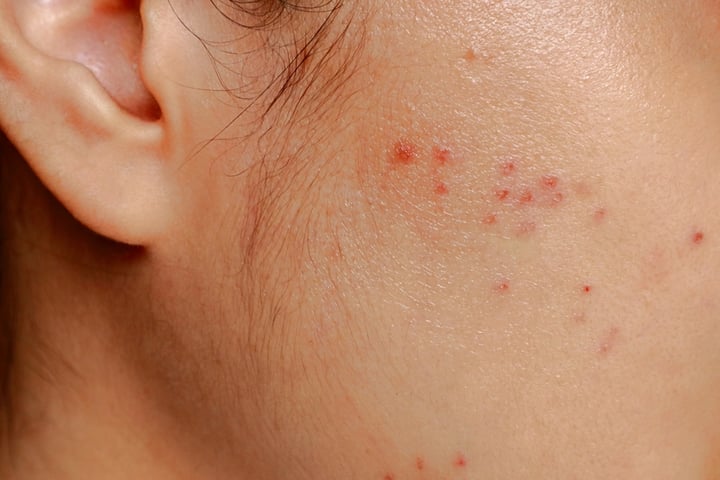
On the same day, the FDA accepted Vanda Pharmaceuticals’ Biologics License Application (BLA) for imsidolimab, a treatment for Generalized Pustular Psoriasis (GPP). This rare inflammatory skin disorder, also known as von Zumbusch psoriasis, is characterized by sudden eruptions of sterile, pus-filled blisters on inflamed skin, often accompanied by systemic symptoms such as fever and fatigue. The FDA has set a target action date of December 12, 2025 for this application.
In pivotal efficacy studies, known as GEMINI-1 and GEMINI-2, a single intravenous dose of imsidolimab led to rapid disease clearance. Notably, 53% of patients achieved clear or nearly clear skin by Week 4, in stark contrast to 13% of those receiving a placebo. The efficacy of imsidolimab was sustained throughout an approximately two-year maintenance period, with no flare-ups occurring in patients receiving the active treatment.
Bysanti Positioned as First-Line Therapy
The FDA’s endorsement of Bysanti positions it as a potential first-line therapy for the acute treatment of manic or mixed episodes associated with bipolar I disorder and schizophrenia in adults. Vanda anticipates that Bysanti will be commercially available by the third quarter of 2026, with marketing exclusivity expected to extend until 2044.
Clinical trials have demonstrated that Bysanti is bioequivalent to iloperidone, leveraging existing efficacy and safety data from Vanda’s previous drug, Fanapt. Fanapt recorded net product sales of $33.2 million in the fourth quarter of 2025, reflecting a 25% increase year-over-year. Furthermore, Vanda is exploring Bysanti’s potential as a treatment for major depressive disorder, with ongoing clinical studies set to conclude by the end of this year.
The broader market saw gains on the previous trading day, with the S&P 500 rising by 0.34% and the Nasdaq gaining 0.42%. Vanda’s stock movement aligns with this positive market sentiment, suggesting that the approval news has positively influenced investor confidence.
Despite this positive news, Vanda Pharmaceuticals is currently trading at 5.5% below its 20-day simple moving average (SMA) and 10.2% below its 100-day SMA, indicating some short-term technical weakness. Over the past 12 months, shares have appreciated by 25% and are closer to their 52-week highs than lows.
The relative strength index (RSI) is at 50.00, indicating a neutral position, while the moving average convergence divergence (MACD) stands at 0.10, slightly below its signal line of 0.15. This combination of neutral RSI and bearish MACD suggests mixed momentum, signaling that traders should exercise caution as the stock adjusts to its current technical environment.
Key resistance levels are noted at $9.50, while support is identified at $8.00. Analysts maintain a Buy Rating for Vanda’s stock, with an average price target of $18.11. Recent analyst actions include HC Wainwright & Co. upgrading their target to $24.00 on February 23, and B. Riley Securities raising their target to $14.00 on December 31, 2025.
At the time of publication, Vanda Pharmaceuticals shares were up 4.53% to $8.53, according to Benzinga Pro data.